Acidity/Basicity Measurement
(Measurement of pH)
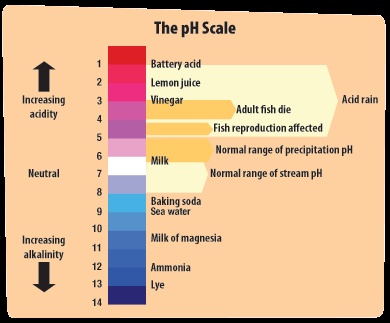
Basicity or acidity is the term extensively used in science field especially in Chemistry. This term is very common in bioscience, environmental science, water treatment, chemical engineering, food technology and nutritional science. It is measured by pH meter, pH-paper (so called litmus paper) and other indicator methods. It is represented by symbol “pH”. It value ranges from 0 to 14. The value from 0 to 6.9 is called acidic and from 7.1 to 14 called basic or alkaline. The solution having pH value of 2 is called strongest acid and of 14 called strongest base. Pure water has pH of 7.
This concept was first introduced by a Danish scientist Soren Peder Lauritz Sorensen. At that time he was working at Carlsberg Laboratory in 1909.

Later his concept was revised in 1924 and represented I term of electrochemical cell. Firstly it was measured by as shown pH but later represented as today.
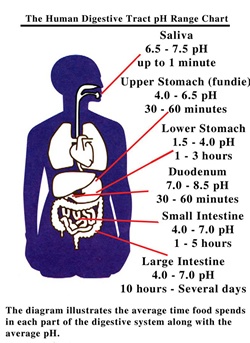
It is recommended to wash mouth after eating meal. It is necessary because acid produce in our mouth due to degradation of sugar and food particles by certain bacteria present in our mouth. It causes the decay of teeth. And this is the Sunnah and also Hadith of Holy Prophet Muhammad P.B.U.H that wash hands after and before taking meal. Our body works best at range of 7.0 to 7.8. Different levels of pH in different parts of human body as shown in figure below.
Ordinary pH of water and other solution can be easily measured by the pH meter. It shown in figure below

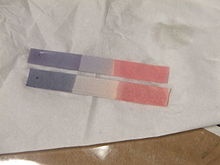
However other non-digital methods are litmus paper method as shown below. It indicates two value i.e red indicates pH less than 4.5 and blue indicate that pH is higher than 8.3.
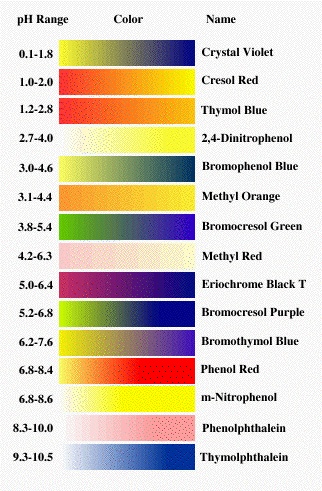
The third way is indicator method. In which a paper is kept in solution and its pH is estimated by matching with standard color as shown below.

The color table is as follows
This was the brief introduction of pH and in next blog it measuring techniques will be discussed. So keep in touch buzz and share.



